-
 Two women suffocate on migrant boat seeking to reach UK
Two women suffocate on migrant boat seeking to reach UK
-
How Schalke returned to the Bundesliga after their 'worst season ever'

-
 Two women die on migrant boat seeking to reach UK
Two women die on migrant boat seeking to reach UK
-
Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'

-
 Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach
Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach
-
Bangkok food vendor curbs push city staple from the streets

-
 More Nepalis drive electric, evading global fuel shocks
More Nepalis drive electric, evading global fuel shocks
-
Latecomer Japan eyes slice of rising global defence spending

-
 Messi goal not enough as Miami collapse in 4-3 loss to Orlando
Messi goal not enough as Miami collapse in 4-3 loss to Orlando
-
German fertiliser makers and farmers struggle with Iran war fallout

-
 OPEC+ to make first post-UAE production decision
OPEC+ to make first post-UAE production decision
-
Massive crowds fill Rio's Copacabana beach for Shakira concert

-
 Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs
Embiid, Maxey shine as 76ers eliminate Celtics in NBA playoffs
-
Fleeting freedom at festival for India's transgender community
-
 Trump says cutting US troop numbers in Germany 'way down'
Trump says cutting US troop numbers in Germany 'way down'
-
Man charged with murdering Indigenous girl in Australian outback

-
 Teen F1 leader Antonelli takes Miami pole as start time moved
Teen F1 leader Antonelli takes Miami pole as start time moved
-
Trump says US not likely to accept new Iran peace proposal

-
 China's Wu Yize wins last-frame thriller to reach snooker world final
China's Wu Yize wins last-frame thriller to reach snooker world final
-
Serene Korda takes three-shot lead at LPGA Mexico

-
 Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux
Golden Tempo wins Kentucky Derby in historic triumph for trainer DeVaux
-
King Charles grasped 'opportunity' on US trip, palace says
-
 China's Wu wins last-frame thriller to reach snooker world final
China's Wu wins last-frame thriller to reach snooker world final
-
Verstappen sees light at the end of tunnel

-
 Young stretches PGA lead to six at Doral
Young stretches PGA lead to six at Doral
-
Rio's Copacabana beach hosts massive crowd for free Shakira concert

-
 Celtics' Tatum ruled out for decisive game seven against Sixers
Celtics' Tatum ruled out for decisive game seven against Sixers
-
Wolff heralds Antonelli speed as teen joins Senna and Schumacher in record books

-
 Senior Iranian officer says fresh conflict with US 'likely'
Senior Iranian officer says fresh conflict with US 'likely'
-
Barcelona on verge of Liga title, Villarreal secure top four

-
 Teen F1 leader Antonelli takes Miami Grand Prix pole
Teen F1 leader Antonelli takes Miami Grand Prix pole
-
Porto edge Alverca to clinch Portuguese league title

-
 US airlines step up as Spirit winds down
US airlines step up as Spirit winds down
-
Barcelona on verge of La Liga title defence with win at Osasuna

-
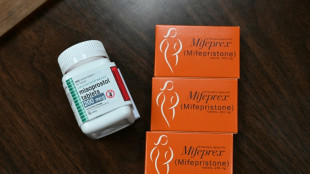 Drugmaker asks US Supreme Court to restore abortion pill access
Drugmaker asks US Supreme Court to restore abortion pill access
-
Schalke return to Bundesliga after three-year absence

-
 NATO, top Republicans question US troop withdrawal from Germany
NATO, top Republicans question US troop withdrawal from Germany
-
Napoli frustrate Como in costly Serie A stalemate

-
 Illegal party at French military site draws up to 40,000 ravers
Illegal party at French military site draws up to 40,000 ravers
-
Arsenal hit stride to go six points clear, West Ham loss offers Spurs hope

-
 Arsenal go six points clear as Gyokeres double sinks Fulham
Arsenal go six points clear as Gyokeres double sinks Fulham
-
PSG fringe team held by Lorient as Bayern Munich return leg looms

-
 Clinical Chennai down Mumbai to keep playoff hopes alive
Clinical Chennai down Mumbai to keep playoff hopes alive
-
Napoli and Como play out goalless draw in Serie A

-
 Murphy into World Snooker Championship final after edging Higgins
Murphy into World Snooker Championship final after edging Higgins
-
PSG held by Lorient with fringe team ahead of Bayern Munich return leg

-
 Aviation companies step up as Spirit winds down
Aviation companies step up as Spirit winds down
-
Champion Norris leads Piastri home in sprint 1-2 triumph for McLaren

-
 UK PM says some pro-Palestinian marches could be banned
UK PM says some pro-Palestinian marches could be banned
-
The Puma out of Kentucky Derby, leaving 19 starters


US health regulator rejects MDMA treatment for PTSD, for now
US health regulators on Friday denied an application for treating post-traumatic stress disorder (PTSD) with the drug MDMA, commonly known as ecstasy, saying more investigation needed to be done.
The company that submitted the application, Lykos Therapeutics, said in a statement that the Food and Drug Administration (FDA) had requested an additional Phase 3 clinical trial to study MDMA's "safety and efficacy."
A panel of experts convened by the FDA to evaluate clinical data on MDMA had overwhelmingly voted in early June to say there was insufficient evidence to prove it was effective.
While unsurprising, the decision announced Friday represents a blow to advocates of the novel treatment.
"The FDA request for another study is deeply disappointing, not just for all those who dedicated their lives to this pioneering effort, but principally for the millions of Americans with PTSD... who have not seen any new treatment options in over two decades, said Lykos CEO Amy Emerson.
PTSD is a debilitating mental health condition that develops after a person experiences or is threatened by traumatic events such as death, combat or sexual assault.
It affects an estimated five percent of Americans in any given year.
Pharmaceutical treatment options for PTSD are so far limited to two antidepressants that require three months of dosing to take effect, and response rates to the medications have been found to be uneven.
MDMA -- methylenedioxymethamphetamine -- is a Schedule 1 drug under the Controlled Substances Act, and approving it for medical use would have represented a major shift.
California-based Lykos based its request for regulatory approval on two clinical studies, each of which enrolled around 100 people, to evaluate MDMA used together with other psychological interventions such as talk therapy, against a placebo with talk therapy.
These two studies, published in the prestigious journal Nature Medicine, indicated MDMA was indeed both safe and highly effective at treating PTSD.
But nine out of 11 experts on the FDA panel said available data was not enough to show the treatment was effective, and 10 out of 11 said the benefits did not outweigh the risks.
In a briefing document put together ahead of the meeting, FDA staff raised concerns about Lykos's clinical trial methodology and criticized the company for not gathering sufficient side effect data.
The company said it will "work diligently in the coming months to address FDA's concerns and to take advantage of agency processes to resolve scientific disagreements."
"We intend to work tirelessly and use all available regulatory pathways to find a reasonable and expeditious path forward," Emerson added.
P.Keller--VB