-
 Ex-F1 driver turned Paralympic champion Zanardi dies
Ex-F1 driver turned Paralympic champion Zanardi dies
-
In Vietnam, Japan PM vows more effort to keep Asia 'free and open'

-
 Humpback whale stranded in Germany released into North Sea: media
Humpback whale stranded in Germany released into North Sea: media
-
Japan PM meets top Vietnam leaders in Hanoi

-
 Spirit Airlines begins 'wind-down', cancels all flights
Spirit Airlines begins 'wind-down', cancels all flights
-
Japan PM to meet top Vietnam leaders in Hanoi

-
 Raisin moonshine banned in Iran enjoys resurgence in New York
Raisin moonshine banned in Iran enjoys resurgence in New York
-
Lebanon says 13 killed in Israeli strikes in south

-
 No.1 Korda charges into share of LPGA Mexico lead
No.1 Korda charges into share of LPGA Mexico lead
-
Young fires 67 to seize commanding PGA lead at Doral

-
 US appeals court temporarily halts mail delivery of abortion pill
US appeals court temporarily halts mail delivery of abortion pill
-
Joy for Norris in Miami as McLaren end Mercedes run

-
 Leclerc offers hope to Ferrari fans in Miami
Leclerc offers hope to Ferrari fans in Miami
-
US to withdraw about 5,000 troops from Germany

-
 'No going back' for Colombia's workers as the right eyes return
'No going back' for Colombia's workers as the right eyes return
-
Norris on sprint pole as McLaren shine again

-
 Venezuelan protesters call government wage hike a joke
Venezuelan protesters call government wage hike a joke
-
Leeds beat Burnley to virtually secure Premier League survival

-
 Gridlock as pandemic treaty talks fail to finish
Gridlock as pandemic treaty talks fail to finish
-
S&P 500, Nasdaq end at fresh records on tech earnings strength

-
 Immersive art: museum-goers in bikinis dive into Cezanne
Immersive art: museum-goers in bikinis dive into Cezanne
-
Gaza activists disperse after flotilla halted by Israel off Crete

-
 US sanctions are 'collective punishment,' says Cuba during May 1 marches
US sanctions are 'collective punishment,' says Cuba during May 1 marches
-
Delhi end slump with team-record chase against Rajasthan

-
 Trump says will raise US tariffs on EU cars to 25%
Trump says will raise US tariffs on EU cars to 25%
-
AI actors and writers not eligible for Oscars: Academy

-
 Rebels take key military base in Mali's north
Rebels take key military base in Mali's north
-
ExxonMobil CEO sees chance of higher oil prices as earnings dip

-
 Leclerc on top for Ferrari ahead of Verstappen and Piastri
Leclerc on top for Ferrari ahead of Verstappen and Piastri
-
Trump says 'not satisfied' with new Iran proposal

-
 After Madonna and Lady Gaga, Shakira set for Rio beach mega-gig
After Madonna and Lady Gaga, Shakira set for Rio beach mega-gig
-
Trump says will raise US tariffs on EU cars, trucks to 25%

-
 Godon raises game to take Romandie stage and revenge over leader Pogacar
Godon raises game to take Romandie stage and revenge over leader Pogacar
-
Celtic's O'Neill expects no let-up from Hibs despite fans' feelings

-
 Pope names former undocumented migrant as US bishop
Pope names former undocumented migrant as US bishop
-
Javelin star Kitaguchi teams up with Czech legend Zelezny

-
 Sawe sub-2hr marathon captured 'global imagination' says Coe
Sawe sub-2hr marathon captured 'global imagination' says Coe
-
King Charles gets warm welcome in Bermuda after whirlwind US visit

-
 Sinner shines to beat Fils, reach Madrid Open final
Sinner shines to beat Fils, reach Madrid Open final
-
UK court clears comedy writer of damaging transgender activist's phone

-
 Was LIV Golf an expensive failure for Saudis? Not everyone thinks so
Was LIV Golf an expensive failure for Saudis? Not everyone thinks so
-
Coe hails IOC gender testing decision

-
 McInnes wants Tynecastle in 'full glory' for Hearts title charge
McInnes wants Tynecastle in 'full glory' for Hearts title charge
-
McFarlane says troubled Chelsea still attractive to potential managers

-
 Man Utd boss Carrick relishes 'special' Liverpool rivalry
Man Utd boss Carrick relishes 'special' Liverpool rivalry
-
Baguettes take centre stage on France's Labour Day

-
 Spurs must banish 'loser' mentality despite injury woes, says De Zerbi
Spurs must banish 'loser' mentality despite injury woes, says De Zerbi
-
Arsenal must manage emotions of title race says Arteta

-
 Nepal temple celebrates return of stolen Buddha statue
Nepal temple celebrates return of stolen Buddha statue
-
US Fed official says rate hikes may be needed if inflation surges


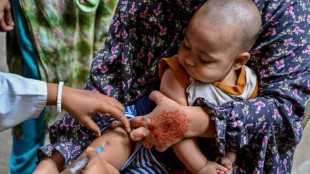
Pfizer to seek US authorization for third Covid shot in children
Pfizer and BioNTech on Thursday announced positive results from a clinical trial on the safety and immune response of a third dose of their Covid vaccine in children aged five through 11, adding they would soon seek regulatory authorization.
Third doses of the vaccine are recommended for those aged 12 and up, and a fourth dose was recently recommended for people over 50.
Younger children -- except for those with immune compromising conditions -- have not been eligible for the third, making them more susceptible to infection from Omicron and its BA.2 subvariant.
BA.2 is now the globally dominant strain, and is behind a current spike in cases in the northeastern United States.
In the phase 2/3 trial, the companies analyzed data from 140 children aged five through 11, approximately six months after the second dose.
The dosage in this group is 10 micrograms, which was selected for safety reasons as children are more susceptible to side effects. The dose for those 12 and up is 30 micrograms.
Across the 140 children analyzed, the third dose was well tolerated, revealing no new safety concerns.
They also analyzed blood sera from a group of 30 individuals, finding that a third dose caused a 36-fold increase in levels of infection-blocking neutralizing antibodies against Omicron, compared to two doses.
Pfizer and BioNTech plan to soon submit the data to the US Food and Drug Administration (FDA), the European Medicines Agency and other regulatory agencies.
Most countries, including the United States, haven't yet authorized Covid vaccines for infants and very young children.
Last month, Moderna said it was pursuing approval for its vaccine in children aged six months through five years, using a two-dose regimen.
Pfizer's vaccine for this group was meant to be considered by the FDA in February but the agency postponed the meeting, because it wanted more data on how it would perform with three doses.
D.Schneider--BTB