-
 Spurs must banish 'loser' mentality despite injury woes, says De Zerbi
Spurs must banish 'loser' mentality despite injury woes, says De Zerbi
-
Arsenal must manage emotions of title race says Arteta

-
 Nepal temple celebrates return of stolen Buddha statue
Nepal temple celebrates return of stolen Buddha statue
-
US Fed official says rate hikes may be needed if inflation surges

-
 Fixture pile-up no excuse for Man City in title race: Guardiola
Fixture pile-up no excuse for Man City in title race: Guardiola
-
Iran offers new proposal amid stalled US peace talks

-
 Gulf countries' plans to bypass Hormuz still far off, experts warn
Gulf countries' plans to bypass Hormuz still far off, experts warn
-
Luis Enrique says 'unique' PSG-Bayern first leg could have gone either way

-
 Rebels take key military camp in Mali's north
Rebels take key military camp in Mali's north
-
Turkish police fire tear gas, arrest hundreds at Istanbul May Day rallies

-
 Lufthansa apologises for lost Oscar after US airport security row
Lufthansa apologises for lost Oscar after US airport security row
-
French hub monitors Hormuz tensions from afar
-
 Flick happy Raphinha back for Barca with title in sight
Flick happy Raphinha back for Barca with title in sight
-
UN troubled by rejected appeal of Cambodian opposition leader

-
 Activists on Gaza aid flotilla detained by Israel disembark in Crete
Activists on Gaza aid flotilla detained by Israel disembark in Crete
-
Oil steady after wild swing, stocks diverge in thin trading

-
 Lufthansa says searching for Oscar lost after US airport security row
Lufthansa says searching for Oscar lost after US airport security row
-
Howe says Saudi backers are fully behind Newcastle

-
 Chinese swimmer Sun Yang reports cyberbullying to police
Chinese swimmer Sun Yang reports cyberbullying to police
-
Solomon Islands leader to face no-confidence vote after appeal court loss

-
 Salah 'deserves big send-off', says Liverpool boss Slot
Salah 'deserves big send-off', says Liverpool boss Slot
-
UK police charge man with stabbing attack on two Jewish Londoners

-
 Solomon Islands leader loses court appeal, must face no confidence vote
Solomon Islands leader loses court appeal, must face no confidence vote
-
Former world skating champion Uno joins pro eSports team

-
 Japan baseball umpire hit by bat still unconscious two weeks on
Japan baseball umpire hit by bat still unconscious two weeks on
-
Nakatani says won't be intimidated in sold-out Inoue title clash

-
 T-Wolves eliminate Nuggets as Knicks demolish Hawks in NBA playoffs
T-Wolves eliminate Nuggets as Knicks demolish Hawks in NBA playoffs
-
Timberwolves eliminate Jokic's Nuggets from NBA playoffs

-
 Arsenal seek to ramp up heat on Man City in title race
Arsenal seek to ramp up heat on Man City in title race
-
PSG closing in on another French title before Bayern second leg

-
 Espanyol must stop rot against Real Madrid as Barca eye title
Espanyol must stop rot against Real Madrid as Barca eye title
-
Leipzig can book return to Champions League as Bundesliga top-four rivals meet

-
 Injuries add to Bath's challenge for Champions Cup semi in Bordeaux
Injuries add to Bath's challenge for Champions Cup semi in Bordeaux
-
Karius getting 'back to the top' with promotion-chasing Schalke

-
 King Charles arrives in Bermuda after whirlwind US visit
King Charles arrives in Bermuda after whirlwind US visit
-
Clashes erupt in Australian town over death of Indigenous girl

-
 Iran war redraws sea routes with Africa as the pivot
Iran war redraws sea routes with Africa as the pivot
-
India's cows offer biogas alternative to Mideast energy crunch

-
 Afghans celebrate spring in bright red poppy fields
Afghans celebrate spring in bright red poppy fields
-
Finland's 'Flamethrower' and 4 other Eurovision favourites

-
 Crude edges up after wild swing, stocks track Wall St rally
Crude edges up after wild swing, stocks track Wall St rally
-
Eurovision: 70 years of geopolitics, patriotism, music and glitter

-
 Knicks demolish Hawks to advance in NBA playoffs
Knicks demolish Hawks to advance in NBA playoffs
-
Blockbuster EU-Mercosur trade deal enters into force

-
 'Uncharted': US court ruling shakes up battle for Congress
'Uncharted': US court ruling shakes up battle for Congress
-
Florida executes man who spent nearly 50 years on death row

-
 Ace lifts rookie Green to share of LPGA lead as Korda lurks
Ace lifts rookie Green to share of LPGA lead as Korda lurks
-
Wear a bulletproof vest? I don't want to look fat, says Trump

-
 Formerra Appoints Matt Borowiec as Chief Commercial Officer
Formerra Appoints Matt Borowiec as Chief Commercial Officer
-
World No. 4 Young leads at PGA Cadillac Championship


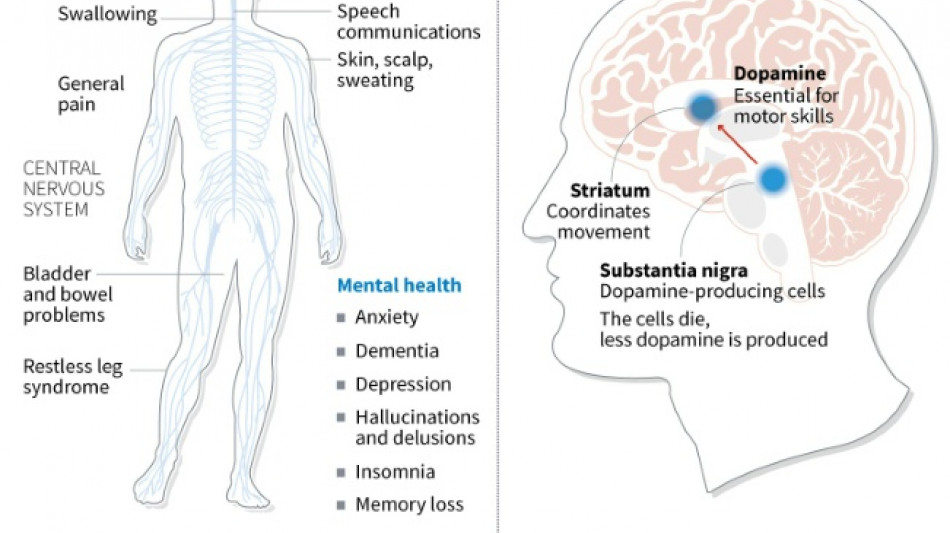
Japan approves stem-cell treatment for Parkinson's in world first
Japan has approved ground-breaking stem-cell treatments for Parkinson's and severe heart failure, one of the manufacturers and media reports said Friday, with the therapies expected to reach patients within months.
Pharmaceutical company Sumitomo Pharma said it received the green light for the manufacture and sale of Amchepry, its Parkinson's disease treatment that transplants stem cells into a patient's brain.
Japan's health ministry also gave the go-ahead to ReHeart, heart muscle sheets developed by medical startup Cuorips that can help form new blood vessels and restore heart function, media reports said.
The treatments could be on the market and rolled out to patients as early as this summer, reports said, citing the health ministry, becoming the world's first commercially available medical products using (iPS) cells.
Japanese scientist Shinya Yamanaka won the Nobel Prize in 2012 for his research into iPS, which have the potential to develop into any cell in the body.
"I hope this will bring relief to patients not only in Japan but around the world," health minister Kenichiro Ueno told a press conference.
"We will promptly carry out all necessary procedures to ensure it reaches all patients without fail."
In a statement, Sumitomo Pharma said it had obtained "conditional and time-limited approval" for the manufacture and marketing of Amchepry under a system which is reportedly designed to get these products to patients as quickly as possible.
The approval is a kind of "provisional license", the Asahi newspaper said, after the safety and efficacy of the treatment was judged based on data from fewer patients than in ordinary clinical trials for drugs.
A trial led by Kyoto University researchers indicated that the company's treatment was safe and successful in improving symptoms.
The study involved seven Parkinson's patients aged between 50 and 69, with each receiving a total of either five million or 10 million cells implanted on both sides of the brain.
The iPS cells from healthy donors were developed into the precursors of dopamine-producing brain cells, which are no longer present in people with Parkinson's disease.
The patients were monitored for two years and no major adverse effects were found, the study said. Four patients showed improvements in symptoms.
Parkinson's disease is a chronic, degenerative neurological disorder that affects the body's motor system, often causing shaking and other difficulties in movement.
Worldwide, about 10 million people have the illness, according to the Parkinson's Foundation.
Currently available therapies "improve symptoms without slowing or halting the disease progression," the foundation says.
iPS cells are created by stimulating mature, already specialised, cells back into a juvenile state -- basically cloning without the need for an embryo.
The cells can be transformed into a range of different types of cells, and their use is a key sector of medical research.
E.Gasser--VB