-
 Spurs must banish 'loser' mentality despite injury woes, says De Zerbi
Spurs must banish 'loser' mentality despite injury woes, says De Zerbi
-
Arsenal must manage emotions of title race says Arteta

-
 Nepal temple celebrates return of stolen Buddha statue
Nepal temple celebrates return of stolen Buddha statue
-
US Fed official says rate hikes may be needed if inflation surges

-
 Fixture pile-up no excuse for Man City in title race: Guardiola
Fixture pile-up no excuse for Man City in title race: Guardiola
-
Iran offers new proposal amid stalled US peace talks

-
 Gulf countries' plans to bypass Hormuz still far off, experts warn
Gulf countries' plans to bypass Hormuz still far off, experts warn
-
Luis Enrique says 'unique' PSG-Bayern first leg could have gone either way

-
 Rebels take key military camp in Mali's north
Rebels take key military camp in Mali's north
-
Turkish police fire tear gas, arrest hundreds at Istanbul May Day rallies

-
 Lufthansa apologises for lost Oscar after US airport security row
Lufthansa apologises for lost Oscar after US airport security row
-
French hub monitors Hormuz tensions from afar
-
 Flick happy Raphinha back for Barca with title in sight
Flick happy Raphinha back for Barca with title in sight
-
UN troubled by rejected appeal of Cambodian opposition leader

-
 Activists on Gaza aid flotilla detained by Israel disembark in Crete
Activists on Gaza aid flotilla detained by Israel disembark in Crete
-
Oil steady after wild swing, stocks diverge in thin trading

-
 Lufthansa says searching for Oscar lost after US airport security row
Lufthansa says searching for Oscar lost after US airport security row
-
Howe says Saudi backers are fully behind Newcastle

-
 Chinese swimmer Sun Yang reports cyberbullying to police
Chinese swimmer Sun Yang reports cyberbullying to police
-
Solomon Islands leader to face no-confidence vote after appeal court loss

-
 Salah 'deserves big send-off', says Liverpool boss Slot
Salah 'deserves big send-off', says Liverpool boss Slot
-
UK police charge man with stabbing attack on two Jewish Londoners

-
 Solomon Islands leader loses court appeal, must face no confidence vote
Solomon Islands leader loses court appeal, must face no confidence vote
-
Former world skating champion Uno joins pro eSports team

-
 Japan baseball umpire hit by bat still unconscious two weeks on
Japan baseball umpire hit by bat still unconscious two weeks on
-
Nakatani says won't be intimidated in sold-out Inoue title clash

-
 T-Wolves eliminate Nuggets as Knicks demolish Hawks in NBA playoffs
T-Wolves eliminate Nuggets as Knicks demolish Hawks in NBA playoffs
-
Timberwolves eliminate Jokic's Nuggets from NBA playoffs

-
 Arsenal seek to ramp up heat on Man City in title race
Arsenal seek to ramp up heat on Man City in title race
-
PSG closing in on another French title before Bayern second leg

-
 Espanyol must stop rot against Real Madrid as Barca eye title
Espanyol must stop rot against Real Madrid as Barca eye title
-
Leipzig can book return to Champions League as Bundesliga top-four rivals meet

-
 Injuries add to Bath's challenge for Champions Cup semi in Bordeaux
Injuries add to Bath's challenge for Champions Cup semi in Bordeaux
-
Karius getting 'back to the top' with promotion-chasing Schalke

-
 King Charles arrives in Bermuda after whirlwind US visit
King Charles arrives in Bermuda after whirlwind US visit
-
Clashes erupt in Australian town over death of Indigenous girl

-
 Iran war redraws sea routes with Africa as the pivot
Iran war redraws sea routes with Africa as the pivot
-
India's cows offer biogas alternative to Mideast energy crunch

-
 Afghans celebrate spring in bright red poppy fields
Afghans celebrate spring in bright red poppy fields
-
Finland's 'Flamethrower' and 4 other Eurovision favourites

-
 Crude edges up after wild swing, stocks track Wall St rally
Crude edges up after wild swing, stocks track Wall St rally
-
Eurovision: 70 years of geopolitics, patriotism, music and glitter

-
 Knicks demolish Hawks to advance in NBA playoffs
Knicks demolish Hawks to advance in NBA playoffs
-
Blockbuster EU-Mercosur trade deal enters into force

-
 'Uncharted': US court ruling shakes up battle for Congress
'Uncharted': US court ruling shakes up battle for Congress
-
Florida executes man who spent nearly 50 years on death row

-
 Ace lifts rookie Green to share of LPGA lead as Korda lurks
Ace lifts rookie Green to share of LPGA lead as Korda lurks
-
Wear a bulletproof vest? I don't want to look fat, says Trump

-
 Formerra Appoints Matt Borowiec as Chief Commercial Officer
Formerra Appoints Matt Borowiec as Chief Commercial Officer
-
World No. 4 Young leads at PGA Cadillac Championship


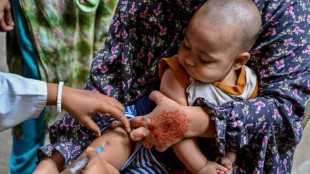
India moves closer to dengue vaccine as final trials underway
As dengue surges globally, an Indian vaccine candidate has entered the final stage of testing, raising hopes for one of the world's first single-dose shots against the deadly mosquito-borne disease.
Dengue, which causes severe flu-like symptoms and debilitating body aches, has exploded globally, fuelled by rising temperatures and densely populated cities.
The World Health Organization (WHO) says that almost half the world's population is now at risk, with 100–400 million infections every year. India alone has recorded over one million cases and at least 1,500 deaths since 2021.
Hoping to stem the global epidemic, Panacea Biotec has begun final Phase III trials of its vaccine, DengiAll, which has been pursuing for nearly 15 years.
More than 10,000 volunteers across the country are enrolled in the study, overseen by the Indian Council of Medical Research, with the vaccine on track for rollout as early as next year if the trial results are favourable.
"We will try to get this vaccine out there as soon as possible," Syed Khalid Ali, chief scientific officer of Panacea, told AFP in New Delhi.
Doctor Ekta Gupta, professor of clinical virology at the Institute of Liver and Biliary Sciences in New Delhi, said dengue was now considered hyperendemic in India, with all four virus serotypes circulating simultaneously.
"This vaccine is very much needed right now to control the occurrence of these cases, or at least prevent the severity."
- Climate change -
Monsoon outbreaks regularly push Indian hospitals to their limits, crowding urban wards and leaving rural regions grappling with late diagnoses and poor access to care.
Higher temperatures and changing rainfall patterns create ideal conditions for Aedes mosquitoes -- the vectors of dengue -- to reproduce and spread the virus.
Children are particularly vulnerable to the more severe form, called dengue hemorrhagic fever, as they are more likely to suffer low platelet counts and shock.
Participants in Phase III trials, which started in 2024, were randomly assigned to receive either the vaccine or a placebo, with the results expected later this year.
Vaccines against all four dengue serotypes have long posed a scientific challenge. Immunity to one strain does not protect against others, and secondary infections can be more severe.
Most existing candidates require multiple doses.
If approved, DengiAll would become one of the world's first single-dose dengue vaccines, following Brazil's approval of a similar shot last year.
It would also be the first such vaccine available in India, where no dengue shot is currently licensed for public use.
"We will be the second (single-dose) vaccine to come out... But in India and several lower-middle-income countries, we will be the first ones to roll out the dengue vaccine," Ali said.
The candidate is based on a tetravalent strain originally developed by the US National Institutes of Health.
- 'Hope for future' -
Panacea is the most advanced of three Indian firms licensed to use the strain, having developed its own formulation and secured a process patent.
Inside the company's research labs, doctor Priyanka Priyadarsiny, head of biological R&D, said vaccine development involves several steps, from proof-of-concept studies to regulatory checks.
"We are extremely cautious about purity, safety and adverse effects," she said. "Only after meeting regulatory specifications can a product be considered safe for public use."
At present, the WHO recommends only one dengue vaccine, Qdenga, produced by Japan's Takeda for children aged six to 16 in high-transmission settings.
Qdenga, which requires two doses administered three months apart, is not currently available in India.
Ali said DengiAll could be given to people aged one to 60 and is expected to offer long-term protection.
In India, final approval would come from the Drug Controller General of India, while WHO prequalification would be required for large-scale international use.
Experts say a successful Indian-made vaccine could be key to affordability and mass rollout in lower-income countries.
Virologist and Oxford University fellow Shahid Jameel -- who is not connected with the trial warned dengue incidence could rise by 50–75 percent by 2050 under current climate change trends.
Still, he cautioned that only Phase III results would determine whether a candidate meets the criteria for a safe and effective dengue vaccine.
"Phase III testing and follow-up are needed to show if the above conditions are met," he told AFP.
"Only then can we have a useful dengue vaccine. It is still early days, but there is hope for the future."
M.Vogt--VB