-
 Ex-F1 driver turned Paralympic champion Zanardi dies
Ex-F1 driver turned Paralympic champion Zanardi dies
-
In Vietnam, Japan PM vows more effort to keep Asia 'free and open'

-
 Humpback whale stranded in Germany released into North Sea: media
Humpback whale stranded in Germany released into North Sea: media
-
Japan PM meets top Vietnam leaders in Hanoi

-
 Spirit Airlines begins 'wind-down', cancels all flights
Spirit Airlines begins 'wind-down', cancels all flights
-
Japan PM to meet top Vietnam leaders in Hanoi

-
 Raisin moonshine banned in Iran enjoys resurgence in New York
Raisin moonshine banned in Iran enjoys resurgence in New York
-
Lebanon says 13 killed in Israeli strikes in south

-
 No.1 Korda charges into share of LPGA Mexico lead
No.1 Korda charges into share of LPGA Mexico lead
-
Young fires 67 to seize commanding PGA lead at Doral

-
 US appeals court temporarily halts mail delivery of abortion pill
US appeals court temporarily halts mail delivery of abortion pill
-
Joy for Norris in Miami as McLaren end Mercedes run

-
 Leclerc offers hope to Ferrari fans in Miami
Leclerc offers hope to Ferrari fans in Miami
-
US to withdraw about 5,000 troops from Germany

-
 'No going back' for Colombia's workers as the right eyes return
'No going back' for Colombia's workers as the right eyes return
-
Norris on sprint pole as McLaren shine again

-
 Venezuelan protesters call government wage hike a joke
Venezuelan protesters call government wage hike a joke
-
Leeds beat Burnley to virtually secure Premier League survival

-
 Gridlock as pandemic treaty talks fail to finish
Gridlock as pandemic treaty talks fail to finish
-
S&P 500, Nasdaq end at fresh records on tech earnings strength

-
 Immersive art: museum-goers in bikinis dive into Cezanne
Immersive art: museum-goers in bikinis dive into Cezanne
-
Gaza activists disperse after flotilla halted by Israel off Crete

-
 US sanctions are 'collective punishment,' says Cuba during May 1 marches
US sanctions are 'collective punishment,' says Cuba during May 1 marches
-
Delhi end slump with team-record chase against Rajasthan

-
 Trump says will raise US tariffs on EU cars to 25%
Trump says will raise US tariffs on EU cars to 25%
-
AI actors and writers not eligible for Oscars: Academy

-
 Rebels take key military base in Mali's north
Rebels take key military base in Mali's north
-
ExxonMobil CEO sees chance of higher oil prices as earnings dip

-
 Leclerc on top for Ferrari ahead of Verstappen and Piastri
Leclerc on top for Ferrari ahead of Verstappen and Piastri
-
Trump says 'not satisfied' with new Iran proposal

-
 After Madonna and Lady Gaga, Shakira set for Rio beach mega-gig
After Madonna and Lady Gaga, Shakira set for Rio beach mega-gig
-
Trump says will raise US tariffs on EU cars, trucks to 25%

-
 Godon raises game to take Romandie stage and revenge over leader Pogacar
Godon raises game to take Romandie stage and revenge over leader Pogacar
-
Celtic's O'Neill expects no let-up from Hibs despite fans' feelings

-
 Pope names former undocumented migrant as US bishop
Pope names former undocumented migrant as US bishop
-
Javelin star Kitaguchi teams up with Czech legend Zelezny

-
 Sawe sub-2hr marathon captured 'global imagination' says Coe
Sawe sub-2hr marathon captured 'global imagination' says Coe
-
King Charles gets warm welcome in Bermuda after whirlwind US visit

-
 Sinner shines to beat Fils, reach Madrid Open final
Sinner shines to beat Fils, reach Madrid Open final
-
UK court clears comedy writer of damaging transgender activist's phone

-
 Was LIV Golf an expensive failure for Saudis? Not everyone thinks so
Was LIV Golf an expensive failure for Saudis? Not everyone thinks so
-
Coe hails IOC gender testing decision

-
 McInnes wants Tynecastle in 'full glory' for Hearts title charge
McInnes wants Tynecastle in 'full glory' for Hearts title charge
-
McFarlane says troubled Chelsea still attractive to potential managers

-
 Man Utd boss Carrick relishes 'special' Liverpool rivalry
Man Utd boss Carrick relishes 'special' Liverpool rivalry
-
Baguettes take centre stage on France's Labour Day

-
 Spurs must banish 'loser' mentality despite injury woes, says De Zerbi
Spurs must banish 'loser' mentality despite injury woes, says De Zerbi
-
Arsenal must manage emotions of title race says Arteta

-
 Nepal temple celebrates return of stolen Buddha statue
Nepal temple celebrates return of stolen Buddha statue
-
US Fed official says rate hikes may be needed if inflation surges


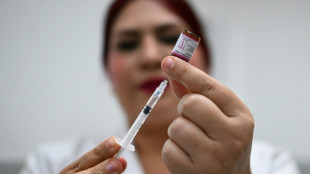
US health experts vote against MDMA as treatment for PTSD
A panel of US health experts convened by the Food and Drug Administration on Tuesday overwhelmingly voted against the use of MDMA, commonly known as ecstasy, to treat post-traumatic stress disorder.
PTSD, a debilitating mental health condition that develops after a person experiences or is threatened by traumatic events such as death, combat or sexual assault, affects an estimated five percent of Americans in any given year.
But pharmaceutical treatment options are so far limited to two antidepressants that require three months of dosing to take effect, and response rates to the medications have been found to be uneven.
California-based Lykos Therapeutics has based its request for regulatory approval on two clinical studies, each of which enrolled around 100 people, to evaluate MDMA used together with other psychological interventions such as talk therapy, against a placebo with talk therapy.
These two studies, published in the prestigious journal Nature Medicine, indicated MDMA was indeed both safe and highly effective at treating PTSD.
But nine out of 11 experts on the panel said available data was not enough to show the treatment was effective, and 10 out of 11 said the benefits did not outweigh the risks.
"I think this is a really exciting treatment. I'm really encouraged by the results to date," said one of the experts, Paul Holtzheimer of the National Center for PTSD.
"But I feel that both from an efficacy and a safety standpoint, it is still premature."
The vote by the panel of experts is non-binding, but the FDA rarely goes against their recommendations.
- Not enough side effect data -
MDMA -- methylenedioxymethamphetamine -- is a Schedule 1 drug under the Controlled Substances Act, and approving it for medical use would have represented a major shift.
In a briefing document put together ahead of the meeting, FDA staff raised concerns that although the studies were nominally "double-blinded" -- meaning neither those being tested nor their health care workers knew who received the treatment versus the placebo -- most people were able to accurately guess what they received.
This "functional unblinding," they argued, introduces bias and uncertainty into study outcomes.
FDA staff also criticized Lykos for not gathering sufficient side effect data, including whether participants experienced "euphoria" or "elated mood," which, they argued, "would be informative for an assessment of abuse potential or characterization of anticipated effects of the drug."
Reports from recreational use suggest MDMA has harmful impacts on heart and liver health -- but the company did not gather enough data in these areas, the FDA said.
- Research misconduct allegations -
Then there were troubling allegations of research impropriety linked to the trials that surfaced in a draft report by the nonprofit Institute for Clinical and Economic Review.
These include claims that the trials "pulled heavily from the existing community of those interested and involved in the use of psychedelics for possible psychological benefits."
Some patients told the nonprofit they were allegedly prevented from entering the long term-follow up study "and felt this was done to keep these negative outcomes out of the data set."
"We certainly take those allegations very seriously and are quite concerned by them," the FDA's Tiffany Farchione said during the meeting, adding a probe was ongoing.
Lykos has said a final decision from the FDA on authorization should come by mid-August.
If the agency were to authorize the treatment over the advisory panel's objections, there could be certain strict stipulations attached -- for example, that the drug only be dispensed in health care settings, with subjects carefully monitored and enrolled in a registry to document side effects.
K.Sutter--VB