-
 Romanian pro-EU PM loses no-confidence motion
Romanian pro-EU PM loses no-confidence motion
-
Stocks diverge as traders eye US-Iran ceasefire

-
 Edin Terzic to become Athletic Bilbao coach next season
Edin Terzic to become Athletic Bilbao coach next season
-
Borthwick backed by RFU to take England to 2027 Rugby World Cup

-
 EU hails 'leap forward' in ties with Russia's ally Armenia
EU hails 'leap forward' in ties with Russia's ally Armenia
-
German car-ramming suspect had mental health problems: reports

-
 Pyongyang calling: North Korea shows off own-brand phones
Pyongyang calling: North Korea shows off own-brand phones
-
Iran warns 'not even started' in Hormuz

-
 World body in dark over allegations against China badminton chief
World body in dark over allegations against China badminton chief
-
Asian stocks drop amid fears over US-Iran ceasefire

-
 China fireworks factory explosion kills 26, injures 61
China fireworks factory explosion kills 26, injures 61
-
China hails 'our era' as Wu Yize's world snooker triumph goes viral

-
 Ex-model accuses French scout of grooming her for Epstein
Ex-model accuses French scout of grooming her for Epstein
-
Timberwolves eclipse Spurs as Knicks rout Sixers

-
 Taiwan leader says island has 'right to engage with the world'
Taiwan leader says island has 'right to engage with the world'
-
Yoko says oh no to 'John Lemon' beer

-
 Bayern's Kompany promises repeat fireworks in PSG Champions League semi
Bayern's Kompany promises repeat fireworks in PSG Champions League semi
-
A coaching great? Luis Enrique has PSG on brink of another Champions League final

-
 Top five moments from the Met Gala
Top five moments from the Met Gala
-
Brunson leads Knicks in rout of Sixers

-
 Retiring great Sophie Devine wants New Zealand back playing Tests
Retiring great Sophie Devine wants New Zealand back playing Tests
-
Stocks sink amid fears over US-Iran ceasefire

-
 G7 trade ministers set to meet but not discuss latest US tariff threat
G7 trade ministers set to meet but not discuss latest US tariff threat
-
Sherlock Holmes fans recreate fateful duel at Swiss falls

-
 Premier League losses soar for clubs locked in 'arms race'
Premier League losses soar for clubs locked in 'arms race'
-
'Spreading like wildfire': Fiji grapples with soaring HIV cases

-
 For Israel's Circassians, food and language sustain an ancient heritage
For Israel's Circassians, food and language sustain an ancient heritage
-
'Super El Nino' raises fears for Asia reeling from Middle East conflict

-
 Trouble in paradise: Colombia tourist jewel plagued by violence
Trouble in paradise: Colombia tourist jewel plagued by violence
-
Death toll in Brazil small plane crash rises to three

-
 Pulitzers honor damning coverage of Trump and his policies
Pulitzers honor damning coverage of Trump and his policies
-
Digi Power X Signs AI Colocation Agreement with Leading AI Compute Company for 40 MW Data Center in Columbiana, Alabama
-
 LA fire suspect had grudge against wealthy: prosecutors
LA fire suspect had grudge against wealthy: prosecutors
-
US-Iran ceasefire on brink as UAE reports attacks

-
 Stars shine at Met Gala, fashion's biggest night
Stars shine at Met Gala, fashion's biggest night
-
Birthday girl, 10, among dead in Colombia monster truck crash

-
 Blake Lively, Justin Baldoni agree to end lengthy legal battle
Blake Lively, Justin Baldoni agree to end lengthy legal battle
-
Dolly Parton cancels Las Vegas shows over health concerns

-
 Wu Yize: China's 'priest' who conquered the snooker world
Wu Yize: China's 'priest' who conquered the snooker world
-
China's Wu Yize wins World Snooker Championship for first time

-
 Broadway theater blaze forces 'Book of Mormon' to close
Broadway theater blaze forces 'Book of Mormon' to close
-
Advantage Arsenal as Man City held in six-goal Everton thriller

-
 Roma hammer Fiorentina to remain in Champions League hunt
Roma hammer Fiorentina to remain in Champions League hunt
-
MLB Tigers star pitcher Skubal to undergo elbow surgery

-
 Oil prices jump on Hormuz tensions as US indices retreat from records
Oil prices jump on Hormuz tensions as US indices retreat from records
-
No.6 Morikawa withdraws from final PGA Championship tuneup

-
 Ukraine and Russia declare separate truces
Ukraine and Russia declare separate truces
-
Arteta warns Atletico will face Arsenal 'beasts' in Champions League

-
 OpenAI co-founder under fire in Musk trial over $30 bn stake
OpenAI co-founder under fire in Musk trial over $30 bn stake
-
Amazon to ship stuff for any business, not just its own merchants


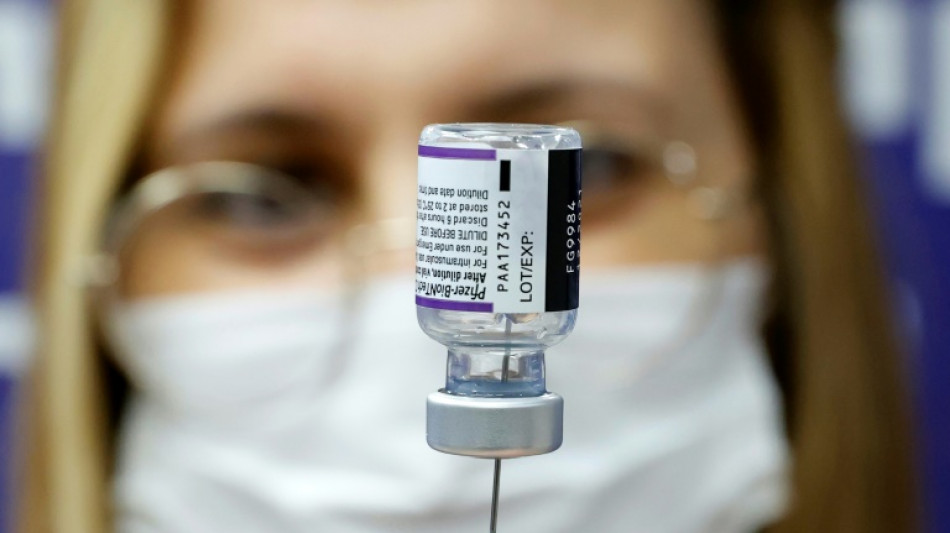
Pfizer Covid vaccine for under-fives effective with three doses
The Pfizer/BioNTech Covid vaccine is safe and effective for children aged six months to under five years when given in three doses, the companies said in a statement Monday.
The announcement comes as the US Food and Drug Administration (FDA) is planning meetings in the coming weeks to weigh authorizing Covid vaccines among the youngest children, the only age group who are not yet eligible in most countries, a source of concern to many parents.
Pfizer/BioNTech evaluated three doses, given at three micrograms, in a clinical trial and found the vaccine evoked a strong immune response. Side effects were similar in the vaccine and placebo groups.
Vaccine efficacy was 80.3 percent, according to a preliminary estimate.
"We are pleased that our formulation for the youngest children, which we carefully selected to be one-tenth of the dose strength for adults, was well tolerated and produced a strong immune response," said Pfizer CEO Albert Bourla in a statement.
"We look forward to soon completing our submissions to regulators globally with the hope of making this vaccine available to younger children as quickly as possible, subject to regulatory authorization," he added.
The FDA has tentatively scheduled three dates in June where experts will meet and likely decide whether to authorize the Pfizer Covid vaccine for under-fives and the Moderna vaccine for under-sixes, which is given as two shots of 25 micrograms.
The agency was originally set to evaluate the Pfizer vaccine given as two doses in February, but data showed it did not provoke a strong enough immune response in children aged two to four. The FDA then asked to see data for a third shot.
- Data welcomed -
According to the new data, 1,678 children received a third dose at least two months after the second dose, at a time when Omicron was the predominant variant.
An analysis of a subset of participants showed antibody levels were similar to 16- to 25-year-olds who were given the full strength vaccine at two doses.
No new adverse events were identified, and the majority of side effects were mild or moderate.
"Three doses of (Pfizer's) Covid vaccine appear to be very safe and highly effective in preventing not only severe disease, hospitalization, and death from Covid, but even symptomatic Covid at a time when Omicron was the dominant variant," Celine Gounder, editor-at-large for public health at Kaiser Health News told AFP.
"However, we know that protection against SARS-CoV-2 infection and milder symptomatic disease wanes over time," added Gounder, an infectious disease specialist and epidemiologist.
"Pfizer is reporting follow-up data only out to seven days after the third dose of vaccine. It's too early to say how the three-dose series would perform out to several months or a year."
Jeremy Faust, of Brigham and Women's Hospital Department of Emergency Medicine, told AFP: "My first impression is very positive. These numbers are exactly the kinds of signals we wanted to see."
"I wish the two-dose series had worked for Pfizer/BioNTech. It didn't. But the three-dose series appears to have given these very young children the protection we want them to have," the doctor added.
If and when both Pfizer's and Moderna's vaccines are authorized, US parents will have to consider whether they want their children to receive Moderna's two dose vaccine -- which will offer faster protection -- or Pfizer's three doses -- which will take longer to be effective but may ultimately be more protective.
Pfizer's announcement comes one week after US health authorities gave the green light for the company's Covid booster shots to be administered to children age five to 11.
Severe disease from Covid is very rare among under-fives but can occur, with 477 US deaths in this age group since the start of the pandemic, or about 0.1 percent of all deaths.
Children can also contract a rare post-viral condition called multisystem inflammatory syndrome in children (MIS-C), which has affected some 8,210 US children and killed 68.
Like adults, some children who get Covid may go on to develop long Covid, with new, ongoing or returning symptoms, including brain fog and fatigue.
I.Meyer--BTB



